Xuan Lia,b,1, Chang Lua,b,c,1, Zhaolin Zhaia,b,1, Robert C. Smithd,1, Suzhen Zhanga,b, Huiling Wange, Chuanyue Wangf, Zhijian Yaog, Zhiyu Chenh, Xiufeng Xui, Shoufu Xiej, Tienan Fengk, Tianhao Gao a,b, Yuke Donga,b,l, Kaiming Zhuoa,b, Qiong Xianga,b, Hua Jinm, John M. Davisn, Kaida Jiango, Yifeng Xua,b,o,q, Stefan Leucht p*, Dengtang Liua,b,l,o,q*
a Division of Psychotic Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, China
b Clinical Center for Psychotic Disorders, National Center for Mental Disorders, Shanghai, China
c Institute of Science and Technology for Brain-Inspired Intelligence, Fudan University, Shanghai, China
d Nathan S. Kline Institute for Psychiatric Research, Orangeburg, New York, and New York University Department of Psychiatry, USA
e Department of Psychiatry, Renmin Hospital of Wuhan University, Wuhan 430060, China
f Beijing AnDing Hospital, Capital Medical University, Beijing 100088, China
g Department of Psychiatry, The Affiliated Brain Hospital of Nanjing Medical University, Nanjing 210029, China
h Department of Psychiatry, The Seventh People's Hospital of Hangzhou, Hangzhou 310013, China
i Department of Psychiatry, The First Affiliated Hospital of Kunming Medical University, Kunming 650032, China
j Department of Psychiatry, The Seventh People's Hospital of Dalian, Dalian 116023, China
k Clinical Research Institute, Shanghai Jiao Tong University School of Medicine, Shanghai 20020, China
l Department of Psychiatry, Huashan Hospital, Fudan University, Shanghai 200040, China
m Department of Psychiatry, University of California at San Diego, San Diego, CA, USA
n Department of Psychiatry, University of Illinois at Chicago, Chicago, IL 60612, USA
o Shanghai Key Laboratory of Psychotic Disorders, Shanghai Mental Health Center, Shanghai JiaoTong University School of Medicine, Shanghai 200030, China
p Department of Psychiatry and Psychotherapy, Klinikum rechts der Isar, School of Medicine, Technical University of Munich, Munich, Germany
q Shanghai Clinical Research Center for Mental Health, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai 200030, China
*Corresponding author:
Professor Stefan Leucht, Klinikum rechts der Isar, School of Medicine, Technical University of Munich, Munich, Germany, e-mail: stefan leucht@tum.de
Professor Dengtang Liu, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, China, e-mail: liudengtang@smhc.org.cn
1 Xuan Li, Chang Lu, Zhaolin Zhai, and Robert C. Smith contributed equally to this work.
While over 50% of patients with first-episode psychosis (FEP) respond to initial antipsychotic treatment, the response rate drops significantly to 34% when patients with an initial treatment failure switch to a second antipsychotic. Ultimately, approximately 20-30% of patients develop treatment-resistant schizophrenia (TRS). Current clinical guidelines worldwide recognize clozapine as the gold standard for treating TRS, yet its use in such patients is often unduly delayed, and it is even rarely prescribed in some countries and regions. Several years ago, European studies suggested that early clozapine initiation (e.g., within 3 months/in patients with initial treatment failure) may improve treatment outcomes, but high-quality randomized controlled trials (RCTs) to support this recommendation have been lacking.
On March 11, 2026, the large-scale, multicenter, randomized controlled Sequential Multiple-Assignment Randomized Trials to Compare Antipsychotic Treatments (SMART-CAT), led by the Shanghai Mental Health Center and jointly conducted by the Beijing Anding Hospital Affiliated to Capital Medical University, Hangzhou Seventh People's Hospital, Renmin Hospital of Wuhan University, Brain Hospital Affiliated to Nanjing Medical University, Dalian Seventh People's Hospital and the First Affiliated Hospital of Kunming Medical University, was officially published in JAMA Psychiatry, a top international medical journal. This study offers new empirical evidence to address the aforementioned clinical challenges.

Adopting a sequential multiple-assignment randomized trial design, the study enrolled 654 patients with FEP who received sequential antipsychotic treatment. Phase 1 was an 8-week RCT, in which participants were randomly assigned to receive olanzapine, risperidone, amisulpride, aripiprazole or perphenazine. A total of 556 patients (85.4%) completed phase 1, among whom 359 achieved a treatment response (defined as a ≥40% reduction in Positive and Negative Syndrome Scale [PANSS] scores), yielding an overall response rate of 55.1%. The response rates across different treatment groups were as follows: risperidone (63.4%), amisulpride (61.8%), olanzapine (60.5%), aripiprazole (44.3%) and perphenazine (45.7%). Comparative analyses revealed that risperidone, amisulpride and olanzapine had significantly higher response rates than aripiprazole and perphenazine.
A total of 111 patients with initial treatment failure proceeded to phase 2, which employed an equipoise-stratified randomization design with participants receiving olanzapine, amisulpride or clozapine. The results showed that the response rate in the clozapine group was 62.5% (20/32), which was significantly higher than that olanzapine group (31.7%, 13/41). Although no significant difference was observed in the response rate between clozapine and amisulpride (44.7%, 17/38), further analysis indicated that the clozapine group had a significantly greater reduction in PANSS scores compared with the control drugs (Cohen d = 0.70; 95% CI, 0.20-1.09; P = 0.005), and results from a mixed-effects model also supported the superior efficacy of clozapine. The findings of this study clearly confirm that clozapine yields the highest response rate when prescribed to patients with initial treatment failure, outperforming olanzapine or amisulpride, and thus provide direct empirical evidence for the early initiation of clozapine.
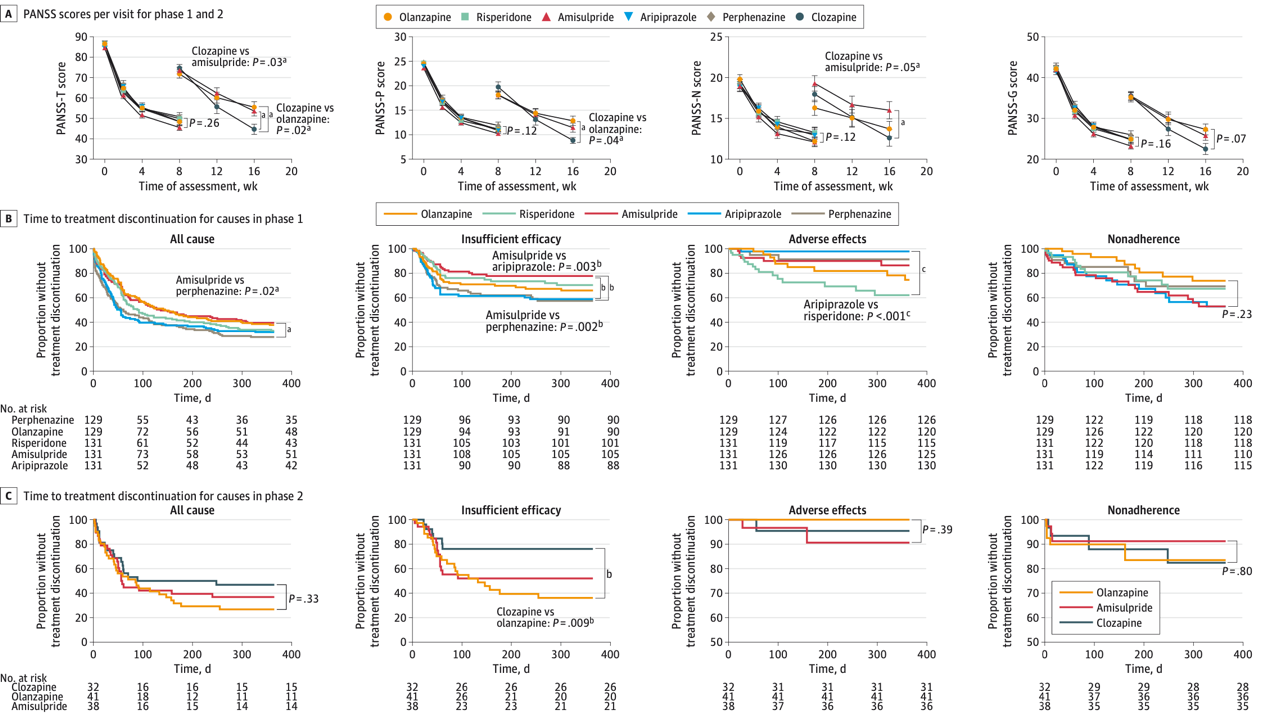
A. Changes in PANSS scores
B & C. Time to all-cause treatment discontinuation
The study also included a 1-year naturalistic follow-up. Although no significant difference was found in the time to all-cause treatment discontinuation between the clozapine group and the control groups, the study still provides valuable evidence-based guidance for clinicians when the clinical goal is to achieve prompt symptom relief and reduce disease chronicity: clozapine should be prioritized as the next treatment option for patients who fail an adequate course of first-line antipsychotic treatment, rather than repeated trials of other first-line drugs.
The mean therapeutic dose of clozapine in this study was 201.85 mg/day, which is lower than the recommended dose in the guidelines of the American Psychiatric Association (APA) but consistent with the conventional dose range (150-300 mg/day) for Asian populations. The results of this study are expected to shorten the trial-and-error period of antipsychotic treatment, improve the overall response rate of schizophrenia treatment, optimize treatment algorithms, thereby reducing the proportion of patients who develop treatment resistance and enhancing the functional prognosis of schizophrenia.
Dr. Xuan Li, Dr. Chang Lu and Dr. Zhaolin Zhai from the Shanghai Mental Health Center, together with Professor Robert C. Smith from the NYU Grossman School of Medicine, are the co-first authors of the study. Professor Dengtang Liu from the Shanghai Mental Health Center and Professor Stefan Leucht from the Technical University of Munich are the corresponding authors. Professor Hua Jin from the University of California, San Diego provided extensive academic support for the study design, interim and final analyses. In addition, the study received guidance and support from Professor John M. Davis, Professor Matcheri S. Keshavan, Professor Stephen R. Marder and Professor Xiaoduo Fan.
Expert Commentary: Reorientation and Future Directions of Clozapine in the Treatment of Schizophrenia
Hua Jin, MD, PhD | University of California, San Diego, USA
Clozapine is one of the most landmark second-generation antipsychotics. Its antipsychotic effect is not primarily based on the strong blockade of dopamine D₂ receptors characteristic of conventional antipsychotics, but rather on the synergistic action of multiple molecular targets including 5-HT₂A, D₄ and α-adrenergic receptors. This unique mechanism enables clozapine to control positive symptoms while also producing modest improvements in negative symptoms and certain cognitive functions. However, its clinical use was once restricted due to the rare risk of agranulocytosis. Despite the improvement of blood monitoring and safety management systems, current clinical guidelines in China, the United States (APA) and the United Kingdom (NICE) yet still recommend clozapine as a third-line agent, to be considered only after the failure of at least two different antipsychotics at adequate doses and durations.
In recent years, systematic reviews and real-world studies have further demonstrated that clozapine consistently outperforms most other antipsychotics in terms of symptom remission rate, duration of sustained remission and reduction in hospital readmission rates. Although its adverse effects such as metabolic syndrome, weight gain and granulocytopenia require regular monitoring, the overall risks are manageable under standardized clinical management.
The study of 654 patients with FEP led by Professor Dengtang Liu from the Shanghai Mental Health Center, published in JAMA Psychiatry (2026), showed that immediate switching to clozapine after failure of the first antipsychotic treatment yields superior efficacy compared with olanzapine and amisulpride. In addition, a network meta-analysis comparing 32 antipsychotics, conducted by the team of Professor Chunbo Li from the Shanghai Mental Health Center in collaboration with German colleagues and published in The Lancet (2026), indicated that clozapine has the best overall efficacy among all antipsychotics evaluated. These findings suggest that the clinical value of clozapine should not be limited to treatment-resistant cases alone, and a more proactive use of clozapine in the early stages of schizophrenia should be considered.
Reference:
1. Li X, Guo X, Fan X, Feng T, Wang C, Yao Z, Xu X, Chen Z, Wang H, Xie S, He J, Zhuo K, Xiang Q, Cen H, Wang J, Smith RC, Jin H, Keshavan MS, Marder SR, Davis JM, Jiang K, Xu Y, Liu D. Sequential Multiple-Assignment Randomized Trials to Compare Antipsychotic Treatments (SMART-CAT) in first-episode schizophrenia patients: Rationale and trial design. Schizophrenia Research. 2021 Apr;230:87-94.
2. Li X, Lu C, Zhai Z, Smith RC, Zhang S, Wang H, Wang C, Yao Z, Chen Z, Xu X, Xie S, Feng T, Gao T, Dong Y, Zhuo K, Xiang Q, Jin H, Davis JM, Jiang K, Xu Y, Leucht S, Liu D. Clozapine After 1 Failed Antipsychotic Drug Trial in First-Episode Psychosis: A Randomized Clinical Trial. JAMA Psychiatry. 2026 Mar 11. doi: 10.1001/jamapsychiatry.2026.0086. Epub ahead of print. PMID: 41811299.
