At the beginning of the new year of 2026, a systematic review led jointly by the team of Professor Chunbo Li from Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, and School of Psychology, Shanghai Jiao Tong University, and the team of Professor Stefan Leucht from Technical University of Munich, Germany, was officially published as an Article in The Lancet. This study compared the efficacy and tolerability of 24 antipsychotic drugs for acute treatment in schizophrenia, representing the largest sample size to date, the most comprehensive coverage of new drugs, and the latest evidence incorporating clinical research from China.
The study included 438 randomized controlled trials with a total of 78,193 participants. It covered 23 antipsychotic drugs primarily acting through dopamine receptor blockade, as well as the novel muscarinic receptor agonist xanomeline-trospium, approved in 2024. The primary outcome was overall symptoms of schizophrenia. Secondary outcomes included positive symptoms, negative symptoms, depressive symptoms, cognitive function, quality of life, social functioning, and tolerability outcomes.
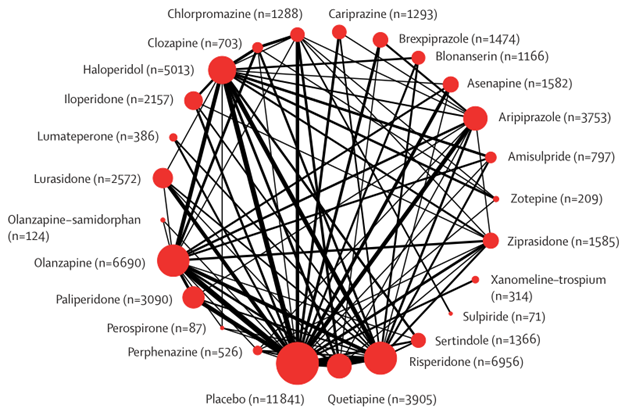
Building upon a 2019 review, this study extended the included antipsychotics to xanomeline-trospium, olanzapine-samidorphan, lumateperone, blonanserin, perospirone, non-oral formulations (long-acting intramuscular or subcutaneous and transdermal delivery), and also included studies with children and adolescents (under 18 years), elderly patients (over 65 years), as well as individuals with first-episode schizophrenia, those unresponsive or intolerant to previous antipsychotic treatment, and those with comorbid drug abuse disorder.
Furthermore, the study provided evidence on 33 outcome measures, including clinically relevant outcomes such as cognitive function, cholinergic adverse events, sexual adverse events, heart rate abnormalities, decreased white blood cell counts, and seizures.
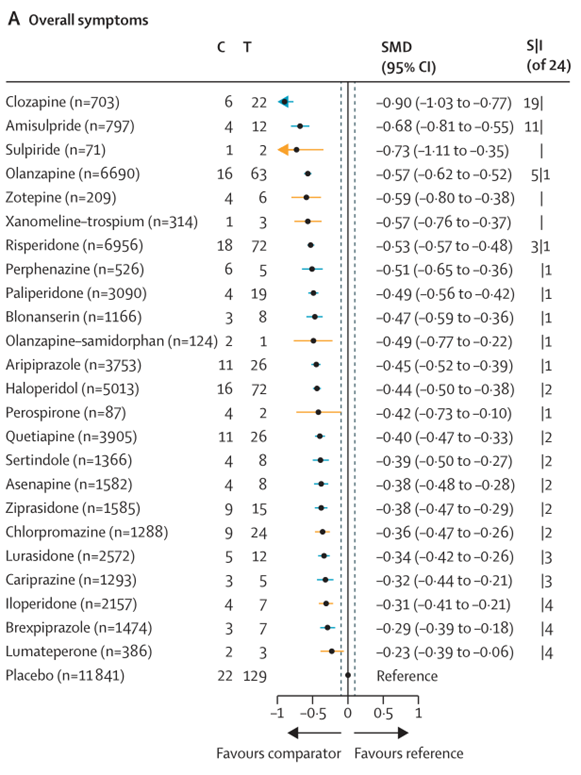
All 24 antipsychotic drugs reduced symptoms more than placebo with standardized mean differences ranging from -0.90 to -0.23. Among them, clozapine, amisulpride, olanzapine, and risperidone were more efficacious than at least three other antipsychotics, and their 95% confidence intervals excluded a "very small" effect size, suggesting these differences are clinically significant. Beyond efficacy, the diverse side effect profiles need be considered for individualized drug choice. Among the partial dopamine receptor agonists, which have generally milder side effects, aripiprazole might be preferred due to its medium-ranked efficacy. Blonanserin showed moderate efficacy and lumateperone was also among those drugs with fewer side effects, but lumateperone ranked last in efficacy. Add-on of samidorphan had little effect on reducing olanzapine-induced weight gain. Muscarinic agents appear to be promising compounds, as xanomeline-trospium ranked in the top third in efficacy and does not cause side effects associated with dopamine receptor blockade, although it does have cholinergic and anticholinergic effects.
For evidence-based medicine, including studies with inappropriate randomization methods and unreliable data can lead to misleading meta-analysis results. Therefore, the Chinese team in this study contacted the first authors and corresponding authors of 5,117 RCTs published on Chinese journals via telephone interviews, emails, and postal questionnaires to obtain detailed information about randomization processing and blinding. However, only 349 studies provided valid responses. Among the studies with returned questionnaires, only 24 Chinese studies confirmed appropriate randomization methods and data reliability, and were ultimately included in the meta-analysis. Although this number is small relative to the total volume of schizophrenia clinical research published in Chinese, this represents the first time such studies have been incorporated into a large-scale systematic review.
In summary, this study provides the most up-to-date and comprehensive comparative evidence on antipsychotic drugs, suggesting differences in efficacy and side effect profiles among various drugs. The findings support more explicit reflection of these differences in clinical guidelines, offering a reference basis for further individualized treatment choices. The study also points out the need for future head-to-head trials, especially comparing xanomeline-trospium with traditional antipsychotics, and research on the use of clozapine in early treatment stages.
Dr. Yikang Zhu, Associate Researcher from Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, and Shanghai Key Laboratory of Psychotic Disorders, is co-first author. PhD students Yu Dong, Shiwei Guan, and Jiaxi Wang are co-authors. The research also received assistance from Professor Xiaochun Qiu (senior librarian) and other teachers and graduate students at Shanghai Jiao Tong University School of Medicine. This study was jointly funded by the National Natural Science Foundation of China International Cooperation and Exchange Program (Grant No. 82161138021) and the German Research Foundation (Grant No. 468853597).
Reference:
Johannes Schneider-Thoma*, Yikang Zhu*, Mengchang Qin, Yu Dong, Shiwei Guan, Jiaxi Wang, Jing Tian, Xiao Lin, Alessandro Rodolico, Spyridon Siafis, Irene Bighelli, Melanie Wehner, Christina Veith, Felix Krayer, Elfriede Scheuring, John M Davis, Josef Priller, Adriani Nikolakopoulou, Georgia Salanti, Chunbo Li†, Stefan Leucht†, Comparative efficacy and tolerability of antidopaminergic and muscarinic antipsychotics for acute schizophrenia: a network meta-analysis of randomised controlled trials indexed in international English and Chinese databases, Lancet 2026; 407: 876--91
